|
However, approaches such as the Ewald sum and the more recent multipole approach have reduced the scaling to approximately linear with no significant loss of accuracy. This would be computationally expensive, as calculations would then scale as the number of particles squared. Unlike the LJ model, which is relatively short-ranged, ionic pair potentials are extremely long-ranged, and interactions between all ions in the system must be considered. Where n i is the number of electrons in the outer shell of ion i, σ i is a parameter related to the size of the ion, and ρ ij is an empirical parameter describing the softness of the electronic cloud. (2) ϕ B - M ( r ) = z i z j e 2 4 π ɛ | r | + A i j ( 1 + z i n i + z j n j ) e ( σ i + σ j − r ) / ρ i j Water molecules form hydrogen bonds with negative ions. Positive ions are attracted to the ion pairs on water molecules and coordinate (dative covalent) bonds may form. It depends on whether there are big enough attractions between the water molecules and the ions to overcome the attractions between the ions themselves. Many ionic solids are soluble in water, although not all. Electron orbital generally does not overlap (i.e., Molecular orbital is not formed), because each of the ions reached the lowest energy state, and the bond is based only (ideally) on the electrostatic interactions between positive and negative ions. Impression of two ions (for example + and −) forming an ionic bond. The larger the difference in electro negativity the more ionic the bond. All bonds have a small amount of covalence. Pure ionic bonding doesnot happen with real atoms. The larger the energy change the stronger the bond. It only occurs if the overall energy change for the reaction is favorable (the bonded atoms have a lower energy than the free ones). Thus, a compound can be classified as ionic or covalent based on the geometry of the atoms. In covalent bonding, the geometry around each atom is determined by valence shell electron pair repulsion theory (VSEPR rules), whereas in ionic materials, the geometry follows maximum packing rules. In an ionic bond, the atoms are bound by attraction of opposite ions, whereas in a covalent bond, atoms are bound by sharing electrons. Such bonds are stronger than hydrogen bonds, but similar in strength to covalent bonds. Ions with opposite charges will attract one another creating an ionic bond. Ionic bonds occur between metals, losing electrons, and nonmetals, gaining electrons. Hans van der Jagt, in Encyclopedia of Analytical Science (Third Edition), 2019 Formation of ionic bondĪn ionic bond can be formed after two or more atoms loss or gain electrons to form an ion. The chloride ion has a − 1 charge because there are 17 protons in the nucleus, but there are 18 electrons around the nucleus of the ion. 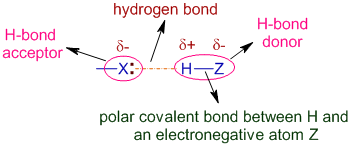
The chlorine atom, which has a high electronegativity, gains an electron and is converted into a chloride ion that has the same electron configuration as argon (1s 2 2s 22p 6 3s 23p 6). It has a + 1 charge, because there are 11 protons in the nucleus, but only 10 electrons around the nucleus of the ion. The resulting sodium ion has the same electron configuration as neon (1s 2 2s 22p 6).  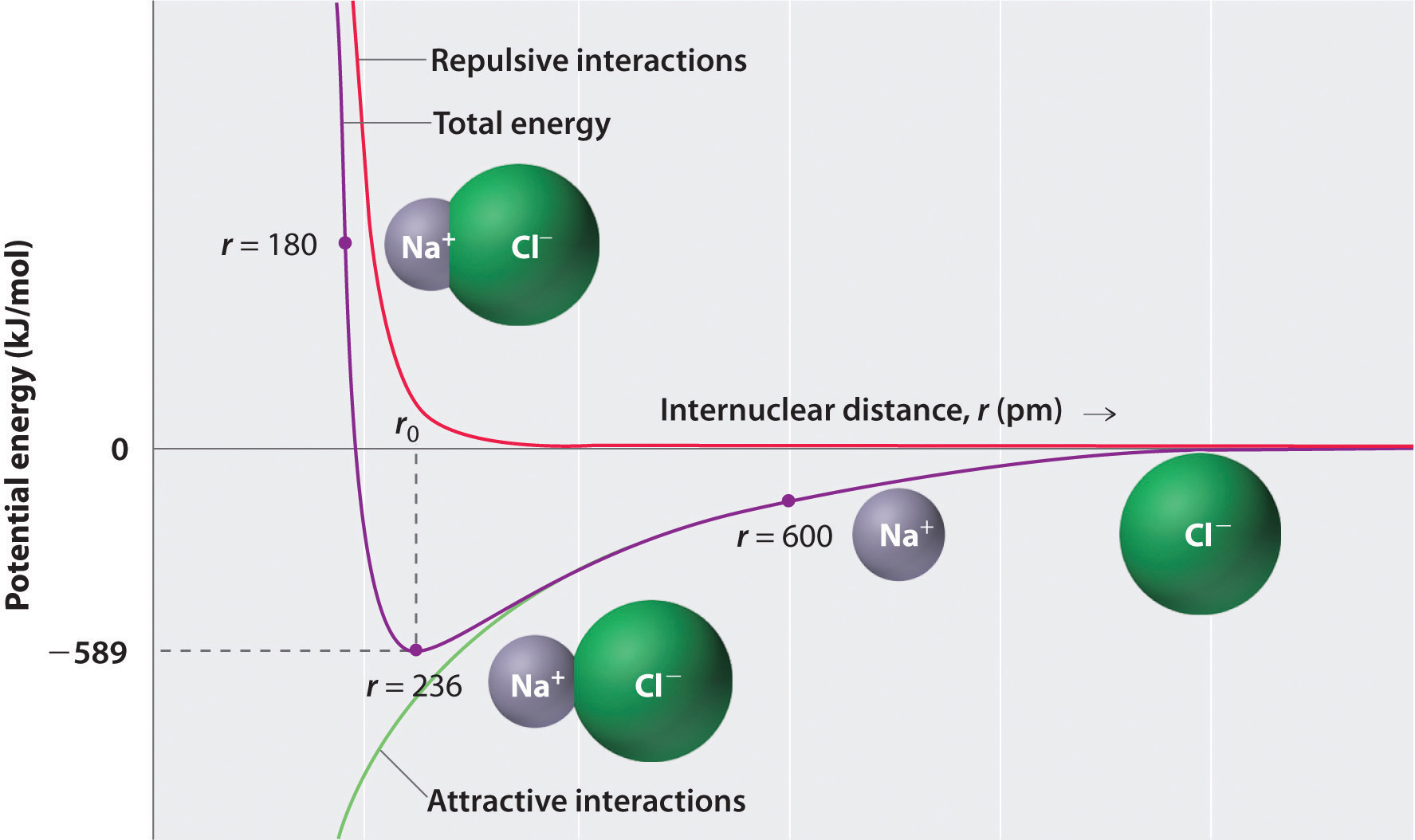 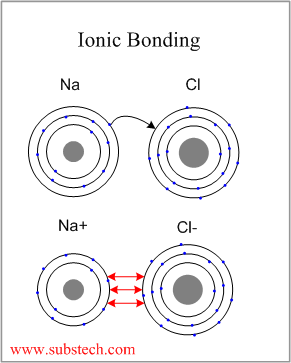
In forming an ionic bond, the sodium atom, which is electropositive, loses its valence electron to chlorine. A chlorine atom, which has 17 protons and 17 electrons, has seven valence electrons in its third shell, represented as 3s 23p 5. A sodium atom, which has 11 protons and 11 electrons, has a single valence electron in its 3s subshell. Sodium chloride is an example of an ionic solid. When the solid dissolves, the ions dissociate and can diffuse freely in solution. Ionic substances exist as crystalline solids. Electron transfer produces negative ions called anions and positive ions called cations. Ionic bonds are formed between two or more atoms by the transfer of one or more electrons between atoms. David Rawn, in Organic Chemistry (Second Edition), 2018 Ionic Bonds
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
